|
From the table above, a single bond between fluorine and nitrogen has a bond length of approximately 64 + 71 =135 pm. Periodic Law states that chemical and physical properties of elements. Basically, what this means is if you drop down a row (period) on the table and move across it, elements follow the same trend as other periods. The bond between fluorine and nitrogen is a single bond. In chemistry, periodicity refers to repeating trends in element properties on the periodic table. To find the nitrogen-to-fluorine bond length in NF 3, draw the Lewis structure. Therefore, the bond length is greater in CO 2.ĥ. There is also a 2019 edition of this table. Here is the pdf file of the color periodic table so you can save and print it. It includes element names, symbols, atomic numbers, atomic weights, element groups, and periods. Chemically, helium behaves like a noble gas, and thus is taken to be part of the group 18 elements. 2013 Edition This free periodic table wallpaper has a white background. They therefore do not follow the octet rule, but rather a duplet rule. Therefore, the bond length is greater in CO 2.Īnother method makes use of the fact that the more electron bonds between the atoms, the tighter the electrons are pulling the atoms together. Period 1 The first period contains fewer elements than any other, with only two, hydrogen and helium. Referring to the table above, a double bond between carbon and oxygen has a bond length of approximately 67 + 57 = 124 pm and a triple bond between carbon and oxygen has a bond length of approximately 60 + 53 =113 pm. Color and black and white tables are given. These high definition images print and resize cleanly. This is a collection of periodic table of elements HD. 
The element name, symbol, number, and atomic mass is easy to read on your device. From the Lewis structures for CO 2 and CO, there is a double bond between the carbon and oxygen in CO 2 and a triple bond between the carbon and oxygen in CO. Germanium, with the chemical abbreviation Ge, is a chemical element having the atomic number 32 and belonging to period 4 of the periodic table of elements. This periodic table of elements is available in a wide range of colors. The bond between carbon and nitrogen is a triple bond, and a triple bond between carbon and nitrogen has a bond length of approximately 60 + 54 =114 pm.Ĥ. To find the carbon-nitrogen bond length in HCN, draw the Lewis structure of HCN. Adding these together and dividing by the number of bonds (3) reveals that the bond order of nitrate is 1.33.ģ. N=O has a bond order of two, and both N-O bonds have a bond order of one. To find the bond order of this molecule, take the average of the bond orders. The Lewis structure for NO 3 - is given below: Mendeleev organized elements according to recurring properties to make a periodic table of elements. 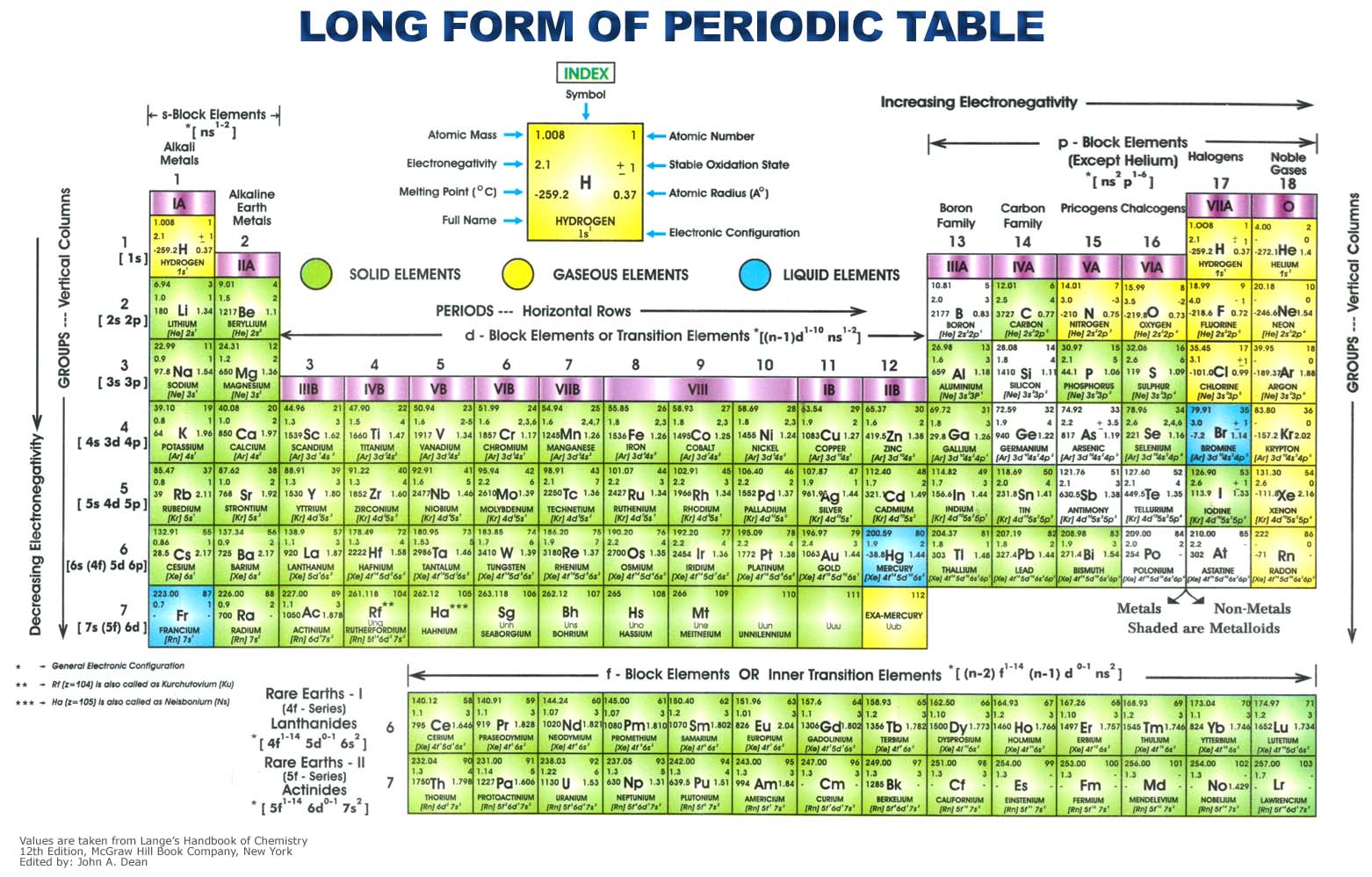
Periodicity is caused by regular and predictable variations in element atomic structure. There is a double bond between the two oxygen atoms therefore, the bond order of the molecule is 2.Ģ. In the context of chemistry and the periodic table, periodicity refers to trends or recurring variations in element properties with increasing atomic number. 
First, write the Lewis structure for \(O_2\).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
